- A PD-L1 test helps doctors determine whether a patient is likely to benefit from immune checkpoint inhibitors.
- Immune checkpoint inhibitors unleash an immune system attack on cancer cells.
A PD-L1 test helps doctors determine whether a patient is likely to benefit from cancer drugs known as immune checkpoint inhibitors. It involves sending a piece of tumor tissue to a lab for analysis.
What is PD-L1?
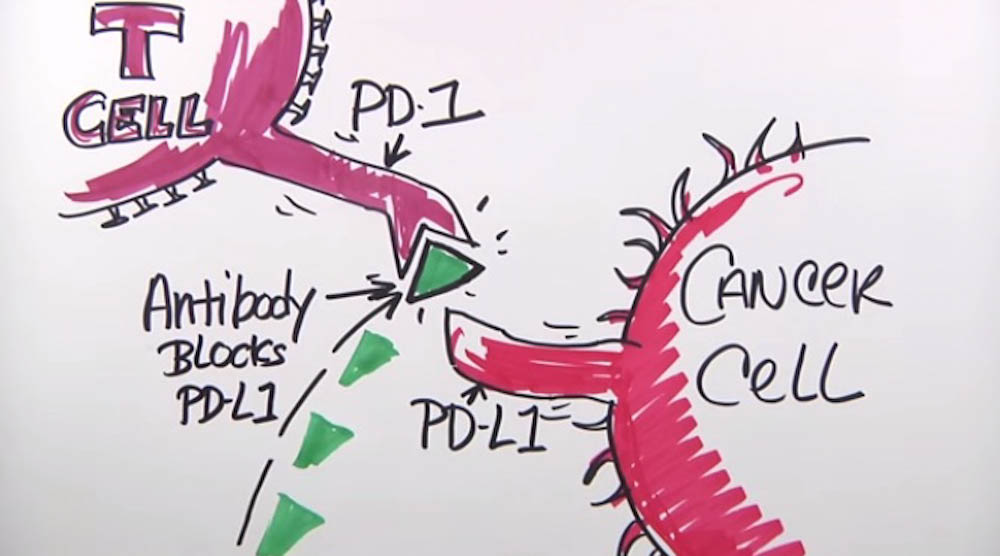
PD-L1 is a protein that allows some cells to escape an attack by the immune system. Extending from the cancer cell surface, PD-L1 interacts with a protein called PD-1 on important immune system cells called T cells. This coupling — known as an immune checkpoint — instructs the T cell to leave the tumor cell alone. Checkpoint inhibitor drugs prevent the PD-1/PD-L1 meeting from taking place. Without receiving the “stop” signal from the PD-L1 protein, the T cells can go ahead an attack the tumor cells.
Which cancers are susceptible to PD-L1?
Drugs inhibiting the PD-1/PD-L1 checkpoint have been approved by the U.S. Food and Drug Administration for several cancers, including:
They are in clinical testing for a variety of other cancer types. A PD-L1 test measures how much PD-L1 a tumor “expresses,” or produces.
Tumors that express high amounts of PD-L1 may be more susceptible to checkpoint inhibitors than those that express less. Patients should ask their cancer physicians whether a PD-L1 test is appropriate for them.
Although helpful in determining which patients may respond to certain drugs, the test is not infallible. Some tumors that test for high levels of PD-L1 may not respond to checkpoint inhibitors, and those that test for low levels may have a strong response. Cancer cells are complex, and a variety of factors can influence how susceptible they are to such drugs.

Neil as you go through your battle. You are being treated by the best in the world, I am praying for a full remission and with the help of God and the Dana Farber I know you will make it.
I have esophogeal cancer and it doesn’t look good for surgery. I went to J.H. in Baltimore a couple weeks ago and I’m interested in the immunotherapy clinical trials as an alternative to the chemo I’ve been getting for more than a year; many, many side effects….
This test was mentioned and I’m going to get that tumor sampled.
Thank you for the good information!