Advances against multiple myeloma have come at an especially rapid pace, with 10 new therapies approved in just the last 10 years. The arrival of a new generation of immunotherapies is accelerating progress even more.
Today, a range of immunotherapies—treatments that marshal the immune system to attack cancer—is available to patients with myeloma. Some are available to certain patients as standard therapy; others are being tested in clinical trials.
As a group, their impact “is greater than anything we’ve seen in myeloma,” says Nikhil Munshi, MD, director of Basic and Correlative Science at Dana-Farber’s Jerome Lipper Center for Multiple Myeloma.

Immunotherapies for myeloma can be divided into three categories: antibody-based treatments, vaccines, and cellular therapies.
Antibody-Based Therapies
Made from cancer-targeting antibodies, some of these therapies are approved for patients whose myeloma has relapsed after previous treatment and these therapies are now being tested in newly-diagnosed patients as well. They have been shown to lengthen the period in which myeloma doesn’t progress, and overall have significantly extended some patients’ survival.
Other antibody-based therapies are under investigation as well. These include antibodies that target specific proteins on myeloma cells; “conjugate” drugs that fuse antibodies to drug molecules in order to deliver the drugs directly to myeloma cells; and “T-cell engagers,” which use antibodies to tow immune system T cells to myeloma cells, prompting an immune attack on myeloma.
Vaccines
Cancer vaccines are designed to spur an immune attack on tumor cells by familiarizing immune system cells with proteins called tumor antigens, which stud the surface of tumor cells. Trials are also examining several myeloma vaccines that use bits of tumor antigens, called peptides, to clue immune system cells in to cancer’s presence. The trials seek to determine if such vaccines can prevent precancerous, or “smoldering,” myeloma from becoming myeloma.
At Dana-Farber, researchers are also participating in a trial of the drug lenalidomide (REVLIMID®) with and without a vaccine, produced from patients’ cancer cells, after they have undergone a transplant using their own stem cells.
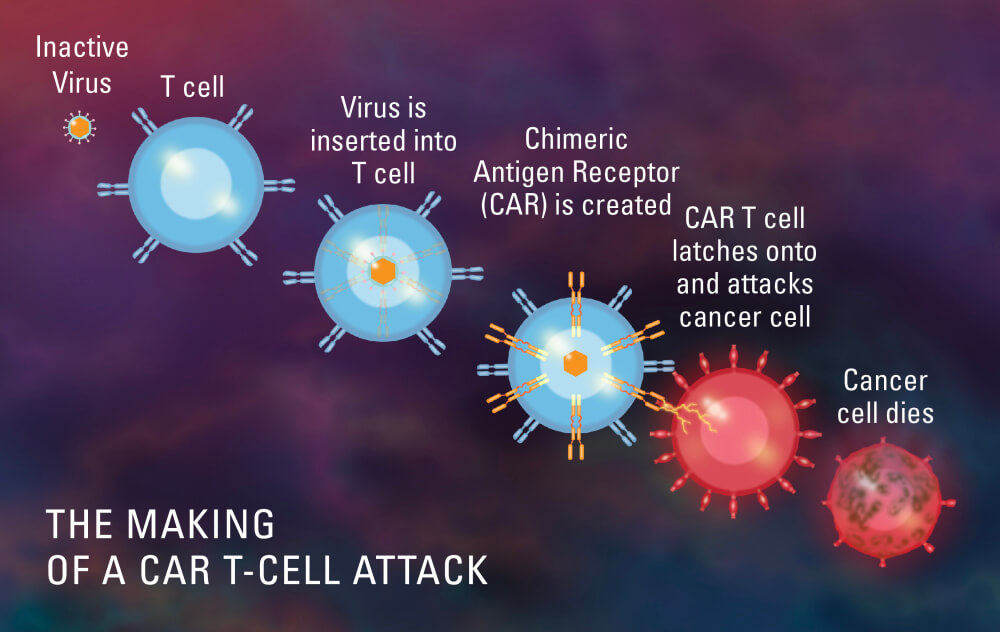
Cellular Therapies
CAR T-cell therapy, a form of cellular therapy in which T cells are genetically engineered to identify and attack tumor cells, has shown striking success in some trials involving patients with myeloma.
In one trial currently underway at Dana-Farber and other centers, a CAR T-cell therapy targeting a myeloma antigen called BCMA produced a response in 95 percent of patients, who all had undergone at least five previous lines of treatment. In a disease where responses to drugs often last a few weeks or months, the average patient response to the CAR T-cell therapy has been nearly a year.
The side effects of immunotherapies are reasonably manageable in most patients with myeloma, Munshi remarks.
As trials continue, researchers are learning which types of immunotherapy perform best in certain patients and certain circumstances. Vaccines may have a primary role in maintenance therapy, which aims to prevent myeloma from returning in patients treated for the disease, Munshi says.
Future clinical trials will explore whether cellular and antibody-based therapies, which are currently being tested in patients who have undergone previous treatments for their myeloma, can be beneficial as earlier-stage treatments, particularly for patients with aggressive forms of the disease.
Learn more about multiple myeloma from Dana-Farber Cancer Institute.

Good to hear.