The wolf at the door is a harmless annoyance compared to the wolf in the house. However dangerous the novel coronavirus is as an infectious agent, it can’t do any harm if it’s locked out of a cell.
Strategies to bar the coronavirus (COVID-19) from entering cells — thereby preventing infection and averting transmission of the virus — are among the most promising treatment approaches to COVID-19. Everything from antibodies to specially made snippets of RNA are being mustered in the effort to develop a safe and effective coronavirus blocker. (RNA is a molecular messenger within cells.)
An approach notable for its ingenuity — and the fact that it has proven successful in other types of viruses — has been pioneered by Loren Walensky, MD, PhD, a pediatric oncologist and chemical biologist at Dana-Farber/Boston Children’s Cancer and Blood Disorders, and his colleagues. Using what are known as stapled peptides, the approach seeks to jam the “landing gear” that the virus uses to fuse to the cells of the human respiratory system.
Creating a ‘decoy’
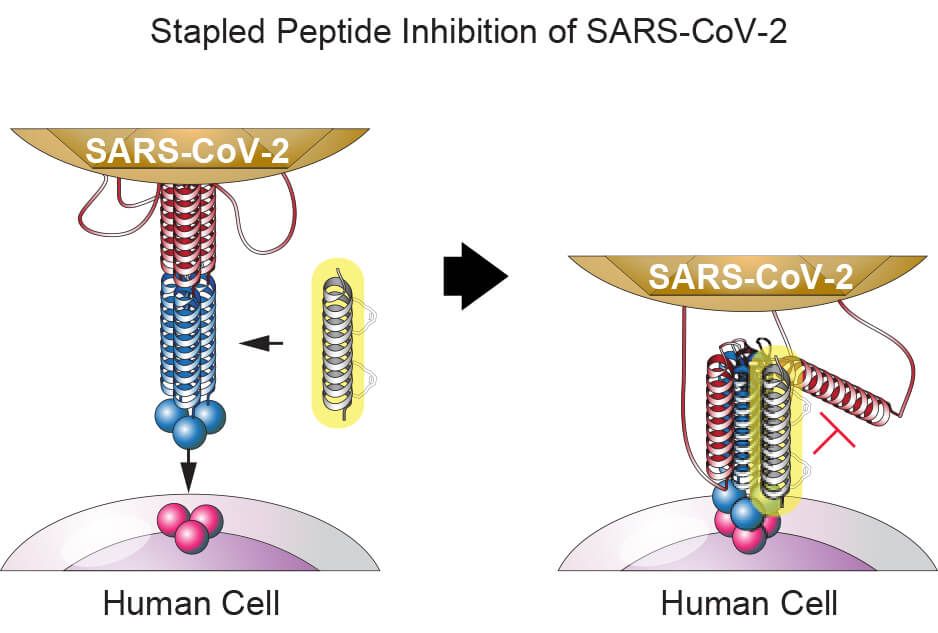
The new virus consists of a short coil of RNA wrapped in a protein envelope that includes stalk-like projections resembling points on a crown. A key surface protein “contains a series of coiled sections known as alpha helices,” Walensky explains. “The virus deploys three of these helices to overlap with three others to form a six-helix bundle that fuses with the human cell membrane, allowing the virus to penetrate and infect the cell.
“Our approach is to make a ‘decoy’ helix that slips between the two groups of helices to prevent the bundle from assembling,” he notes.
The decoy consists of a stapled peptide. Peptides are short chains of amino acids — not long enough to be full-fledged proteins — but can interact with proteins to modulate a variety of cellular functions.
“One advantage of using peptides is that they are nature’s solution to targeting proteins,” Walensky remarks.
A ‘staple’-based approach
Early attempts to use natural peptides as therapies faced obstacles because the peptides can lose their shape and get degraded rapidly once injected into the body.
A chemistry-based solution was to reinforce peptides with hydrocarbon “staples.” In 2010, Walensky and his associates showed that a stapled peptide could effectively target the fusion apparatus used by HIV-1, the virus that causes AIDS.
Because many other viral families — including those of the RSV, SARS, MERS, and Ebola viruses, as well as the novel coronavirus — use the same fusion mechanism, there are valid reasons for believing this approach will work against them as well.
“As we previously demonstrated for the respiratory syncytial virus (RSV), our newly designed stapled peptides targeting the novel coronavirus’ helical bundle may be effective both as a way to prevent infection as well as to block the spread of infection to the lungs in people with established infection,” Walensky comments.
Experiments are currently underway to test the Walensky lab’s peptides against the novel coronavirus that causes COVID-19.
