- Natural killer (NK) cells are white blood cells that have specialized roles as part of the body’s immune response and can kill tumor and infected cells through direct contact.
- While NK cells are potent first responders to infection and disease, they represent only a small proportion of all white blood cells and don’t endure for very long.
- Scientists are working hard to boost the cells’ effectiveness and persistence as NK cells have shown potential for use in cancer immunotherapy, though clinical application is still largely experimental.
Natural killer (NK) cells are white blood cells that serve as a part of the body’s first line of defense against infections and cancers. Along with T cells and B cells, NK cells have specialized roles as part of the immune system and have shown potential for being used in cancer immunotherapy. Their effectiveness is still being investigated; the use of NK cells in cancer treatment is not yet approved by the U.S. Food and Drug Administration.
Researchers at Dana-Farber have made key advances in the effort to harness NK cells for cancer treatment. In the 2010s, Rizwan Romee, MD, found a way to prime NK cells so they live longer and form memories of the cancerous cells they’ve encountered — critical qualities for success as a cancer therapy.
Today, Dana-Farber investigators are national leaders in the development and testing of NK cell therapies for a wide range of cancers. They’re leading clinical trials of the therapies in patients with multiple myeloma or head and neck cancers, and in patients with acute myeloid leukemia, myelodysplastic syndromes, or myeloproliferative neoplasms who have relapsed after a haploidentical hematopoietic stem cell transplant.
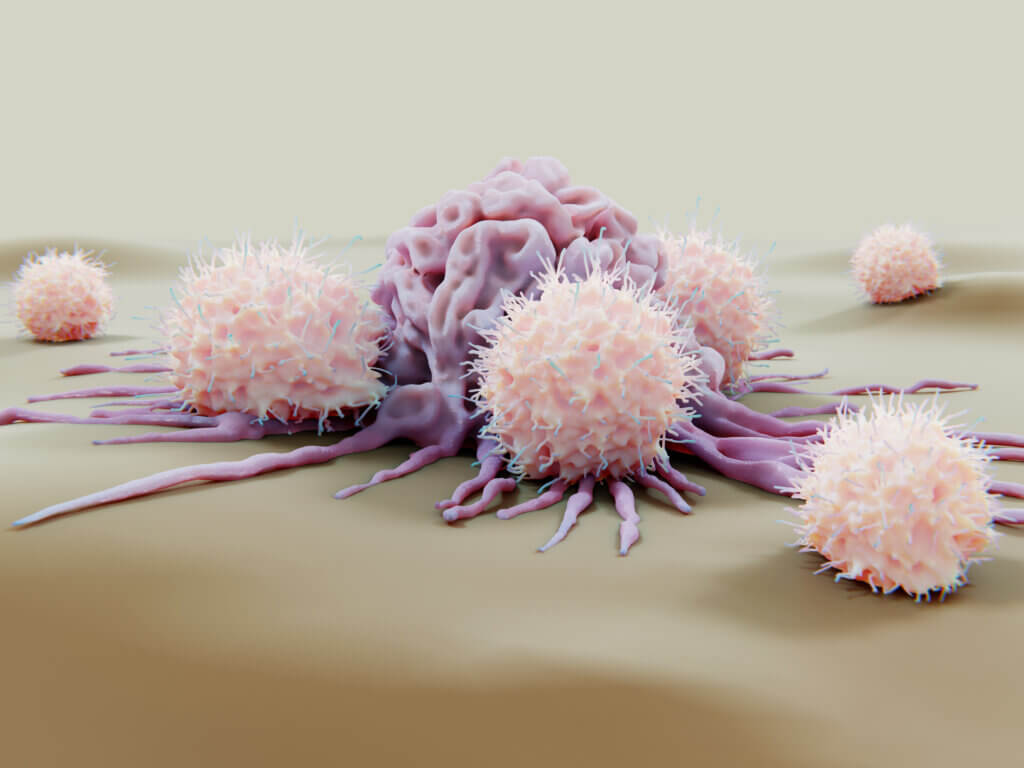
How NK cells work
NK cells are potent first responders that can kill tumor and infected cells by direct contact, and they also can help regulate the immune system. Unlike T cells and B cells, NK cells can recognize and kill abnormal cells without ever having encountered the foreign cells or having had the foreign cells “presented” to them for recognition by MHC (major histocompatibility locus) molecules. However, NK cells make up a relatively small proportion of white blood cells, and they don’t endure for very long. Scientists are working hard to boost their effectiveness and endow them with “memory-like” powers for greater persistence.
T cells, on the other hand, make up a much larger part of the immune army. They aren’t first responders but go on the attack in a delayed fashion after having been primed to recognize specific identifying molecules, or antigens, on infected or cancerous cells. They also persist longer in the body. T cells are the immune soldiers that are unleashed against cancer by the checkpoint blocker drugs like pembrolizumab, nivolumab and ipilimumab.
“The unique capabilities of NK cells and T cells complement each other,” Romee says. “They make it possible for the immune system to recognize a broader array of diseased cells than it could with either NK or T cells alone.
“Infected or cancerous cells oftentimes have a certain group of molecules on their surface yelling for help, saying, basically, ‘We are infected, we are malignant, we have stress in our nucleus or genes.’ NK cells respond by becoming activated and killing the cells to prevent the infection or disease from spreading,” Romee explains.
NK cells also have an advantage in that they are relatively safe and less likely to cause graft versus host disease (GVHD) — where white blood cells present within the transplanted tissue attack the recipient’s body cells.
What’s the latest in NK cell research?
Along with other forms of immunotherapy, there is a great deal of research on how NK cells could be used to treat cancer.
In general, NK cells are harvested from healthy donors and activated by exposing them to various chemical signaling substances; they are then infused into the patient. There is a big push towards engineering NK cells that better target cancer and persist longer in the body after being infused.
A new clinical trial for patients with head and neck cancers is testing an innovative way to accomplish that. It’s a follow-up to a previous trial that used “haploidentical” NK cells from first-degree relatives of study participants. (Such cells are a partial immunological match to a patient’s own cells.) The results of the trial are currently being analyzed.
The new, phase 2 trial involves several major changes in the treatment protocol. For one, patients in the earlier trial received large doses of chemotherapy create room for the donated cells in the bone marrow. Such high doses can be difficult for patients to tolerate and can raise the risk of infection – plus, the bone marrow doesn’t always recover well. The trial also involved just a one-time infusion of donor cells. The cells would initially be active against the cancer but lost their potency over time.
The follow-up trial, which is now open, solves many of those issues. First, it doesn’t require a specific donor for each patient: the NK cells used in the trial are an “off-the-shelf product” — specially engineered to be effective in all participants.
“The cells were ‘immortalized’ in a lab so they can proliferate indefinitely and were genetically modified to express the PD-L1 receptor [a structure on the cells that binds to a corresponding protein on tumor cells],” says Dana-Farber’s Glenn J. Hanna, MD, who is leading the trial. “They include the best features of T cells and NK cells.”
Patients do not have to undergo chemotherapy prior to receiving the modified NK cells. The cells also can be given multiple times: patients participating in the new trial will receive them twice a month. On the same day they are infused with the cells, patients will receive a growth factor injection to help the cells persist longer in the body. Patients will also receive an antibody-based drug called cetuximab, which, on its own has effectiveness against head and neck cancers by targeting NK cells. Investigators plan to enroll about 25 patients in the trial.
Romee has been using NK cell-based therapy to treat patients who have relapsed after having had an haploidentical (half-matched donor) stem cell transplant for a blood cancer. A number of such patients received adoptively transferred memory-like NK cells followed by seven doses of interleukin-2 to stimulate the expansion of NK cells. In the first six patients of the phase 1 trial there was a 10 to 50-fold expansion of NK cells that was sustained over months. Romee commented that these NK cells “serve as a promising platform for the treatment of post-transplant relapse of myeloid disease.”
About the Medical Reviewer

Dr. Hanna completed his fellowship training in hematology and medical oncology at Dana-Farber Cancer Institute in 2016. Prior to this, he earned his medical degree from Georgetown University School of Medicine in 2010, where he graduated summa cum laude, a member of Alpha Omega Alpha Honor Society and the Kober Medalist for academic excellence. He joined the faculty of the Center for Head and Neck Oncology in 2017. Dr. Hanna is the Director of the Center for Cancer Therapeutic Innovation (CCTI), the early drug development program at DFCI. His clinical and translational research efforts focus on precision medicine approaches to treat head and neck cancers. He has special interests in salivary gland cancers and rare head and neck malignancies, and in molecular and immunologic biomarker discovery.

I appreciate you providing this information about natural killer cells and how they combat cancer. For me, the information is extremely helpful.