SUMMARY: Researchers are learning how best to use this powerful modality while avoiding serious toxicities.
Since it was approved by the U.S. Food and Drug Administration (FDA) in the fall of 2017, a form of the powerful and promising therapy known as CAR T-cell therapy has been used to treat certain young patients with B-cell acute lymphoblastic leukemia (ALL) who have relapsed or didn’t respond to standard regimens.
Today, researchers and physicians at Dana-Farber/Boston Children’s Cancer and Blood Disorders Center say they are learning how best to use the specific CAR T-cell treatment known as Kymriah — for which patients, and at what point in their cancer treatment — and how to predict and treat the serious toxicities that can occur. They’re also laser-focused on another question: Can CAR T-cell therapy work for other pediatric patients?
A paradigm shift
CAR T-cell therapy is a form of gene therapy in which a patient’s own immune cells are removed, engineered in a lab to hone their cancer-fighting abilities, and returned to the body. In clinical trials, Kymriah showed encouraging remission and survival rates in B-cell ALL patients who had few other options — leading to its FDA approval.
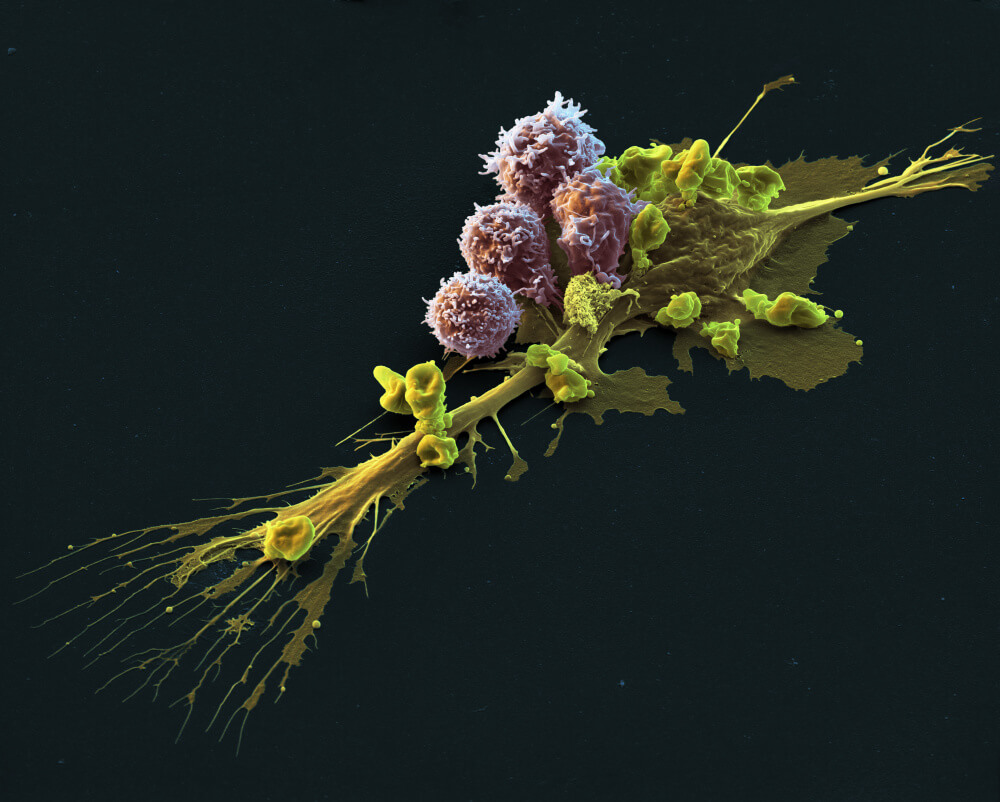
Kymriah is approved for patients 25 years old and below who have B-cell ALL expressing the CD19 protein, and who have not responded to standard therapy or are in a second or later relapse. The great majority of patients with B-cell ALL are successfully treated with standard methods, but those who don’t respond have an unfavorable prognosis. Kymriah has been shown to be effective in many of these cases.
“CAR T-cell therapy is a complete paradigm shift,” says Christine Duncan, MD, a senior physician at Dana-Farber/Boston Children’s. “It started with CD19, which is a specific form of pediatric ALL. Now that it has expanded, we’re learning a lot more about the obstacles to therapy and how we need to pick the right patients for the treatment, so we can move forward to other high-risk populations.”
Order of operations
Some patients receive CAR T-cell therapy and then undergo stem cell transplantation, while for others it’s the reverse, say Duncan and Steven Margossian, MD, PhD, who is a senior physician in the stem cell transplant program at Dana-Farber/Children’s.
“Generally, we are using CAR T as a bridge to a transplant; CAR T gets the patient into a good remission and then we take them to transplant,” says Margossian. “They do well with a transplant.”
But some patients whose leukemia relapses after a stem cell transplant are treated with Kymriah as definitive therapy.
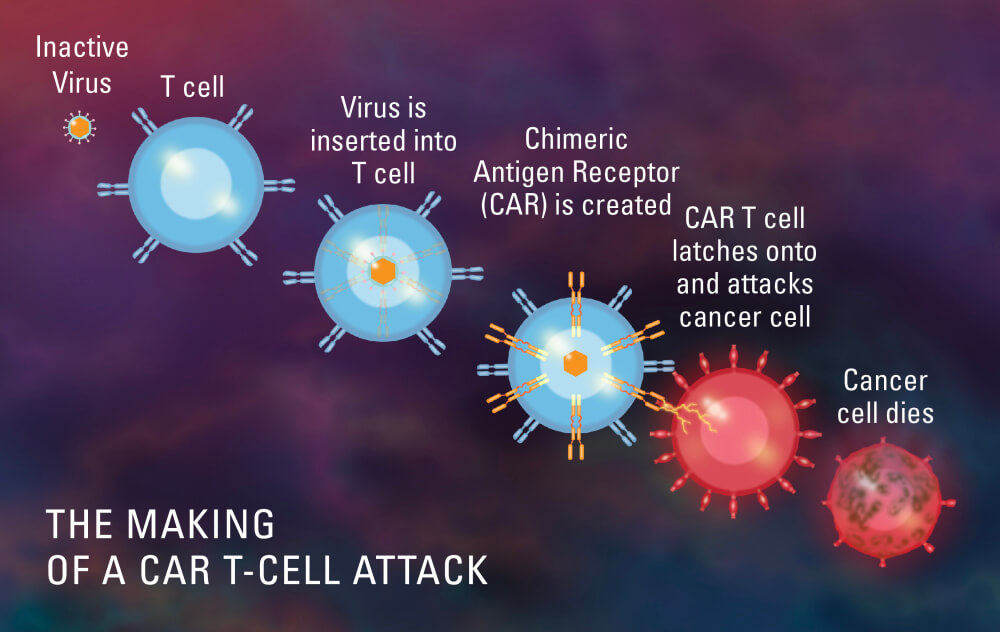
Kymriah treatments are custom-made for each individual. Initially, the patient undergoes apheresis to remove lymphocytes known as T cells, which are frozen, packaged, and sent to a pharmaceutical laboratory. There, the T cells are equipped with what’s known as a chimeric antigen receptor (CAR) so that they will home in on the CD19 protein on the patient’s leukemia cells. This takes about 24 to 28 days; meanwhile, the patient may receive a cycle of chemotherapy to combat the leukemia. When the CAR T cells are returned to Dana-Farber/Boston Children’s, they are reinfused into the patient, where they seek out and destroy the cancer cells.
Side effect management
CAR T-cell therapy can trigger serious side effects, including cytokine release syndrome. There are medications that can block this reaction, “and we are researching the correct way to give the medication: should it be as a rescue medicine or can you give it preemptively?” Margossian says. Neurological toxicities are also possible.
“We are very good at managing the patients through the complications — we always have regular meetings for every CAR T patient that comes in, and we are always prepared to transfer the patient to the ICU if needed,” Margossian notes.
The long-term effects of CAR T-cell therapy are also still unclear, but doctors are working to understand them.
“It took many years to figure out how to correctly follow patients who received a stem cell transplant,” says Duncan. “Now we’re trying to figure out how to follow patients who received CAR T-cell therapy. This is something we’ll learn over time.”
Meanwhile, variations on the approved use of Kymriah are being studied in new clinical trials. Margossian says one trial will be for patients with ALL who are in remission but have high levels of detectable cancer cells. Another trial will test Kymriah in pediatric B-cell lymphomas. Also on the horizon is a clinical trial of CAR T-cells as a bridge to transplant for patients with acute myeloid leukemia (AML).
Farther off are CAR T-cell therapy trials for solid tumors such as bone cancers and neuroblastoma, which present unique challenges, according to Margossian. One hurdle is that the cancer-specific molecules that CAR T cells bind to are often inside the cancer cells of solid tumors, not on the surface, as they are in blood cancers, and therefore are more difficult to target.
“It’s exciting to see CAR T-cell studies developing and expanding to very high-risk patient populations,” says Duncan. “We’re very hopeful that CAR T-cell therapy will expand to entirely new populations.”
