When Dana-Farber’s Catherine Wu, MD, was a newly minted physician in the mid-1990s, there was just one major form of immunotherapy for cancer — and it had been designed with an entirely different purpose in mind.
Bone marrow transplantation, which was first developed in the 1970s, was conceived as a way of dealing with the effects of high-dose chemotherapy for leukemia, lymphoma, and other blood-related diseases. The large doses killed diseased blood cells throughout the body but also destroyed the bone marrow, birthplace of new blood cells. By transplanting bone marrow from a compatible donor, doctors could restore what had been lost during treatment.
As time went on and physicians gained experience with the procedure, it became clear that transplanted marrow was far more than just a “replacement part.” The new marrow, like the old, contained stem cells that generated the major components of the blood supply: red cells to carry oxygen, platelets to aid in clotting, and white cells to fend off disease.

But doctors and researchers quickly realized that the donated marrow was itself a potent weapon against leukemia. The brood of new white blood cells that pour into the bloodstream attack tumor cells that haven’t succumbed to chemotherapy, producing lengthy remissions, even cures in many patients.
In a way its originators could barely have imagined, bone marrow transplantation (and, more recently, stem cell transplantation) essentially involves endowing a patient with a new, more nimble immune system. The action of the transplanted tissue upon the disease came to be known as the graft-versus-leukemia (GVL) effect.
Wu, whose research in the genomics of chronic lymphocytic leukemia (CLL) led to her work on a vaccine that rouses the immune system to battle cancer, was introduced to the graft-versus-leukemia effect as an intern at Brigham and Women’s Hospital. It would set the course of her career.
“As an MD, my first interest is in patients, how they go through therapy and how we can develop cures,” she relates. “And I’ve always been fascinated by the concept that the immune system can recognize malignant cells as something that should not be in the body. Stem cell transplantation was the first example in humans of a treatment that could trigger an anti-tumor immune response. When I learned about graft-versus-leukemia, I thought, ‘This is it, this is going to be my field.’”
Part of the appeal of transplantation was its ability to deliver, for some patients, the most desired of outcomes: a cure.
“Knowing that we had patients in the clinic who had been treated more than a decade previously and were doing well was enough proof to me that this was a path worth focusing on,” Wu comments.
Impressive as transplantation’s clinical results can be, Wu was equally intrigued by the biology behind them. By the early 2000s, scientists had a basic understanding of the GVL effect, but many details remained obscure. By beginning with a therapy she knew to be effective, Wu could probe why it was effective and use that knowledge in developing other forms of cancer immunotherapy.
“My question was, if we’re seeing responses after an infusion of donor cells, what exactly do cells ‘see’ when they enter the body, and how does that effect their interaction with tumor cells?” Wu remarks. “Is there toxicity? Is there specific eradication of the malignant cells?”
Answering questions like these — uncovering what Wu calls the “ingredients” of the GVL effect — might make it possible to harness the immune system in other therapies that don’t involve the extraordinary complexity and preparation involved in stem cell transplants.
Pioneering spirit
When Wu made her decision to explore the immunological intricacies of stem cell transplantation, cancer immunology was not the star attraction it has since become. The recognition of the GVL effect had blown a hole in the once-widespread belief that the immune system had little effect on cancer, but many scientists remained skeptical about immunotherapy’s ability to thwart the disease.
Wu’s desire to enter the field was, if not radical, certainly unconventional.
“I’ve always been fascinated by the concept that the immune system can recognize malignant cells as something that should not be in the body.“
Catherine Wu, MD
“Scientists are, as a rule, creatures of passion,” Wu relates. “We do this because we really believe in something and want to understand what’s going on. Harnessing immunity made sense to me, as it made sense to others who chose this as their area. Immunology is complex; it requires coordination between many different cell types of different types of tissue. GVL was a clue to how effective the immune system could be; that’s what I wanted to pursue.”
When Wu set up her laboratory at Dana-Farber in 2005, her research centered on not only GVL but also the genomics of CLL — the spectrum of mutations and other genetic abnormalities within CLL cells and, particularly, how those mutations change over time, as the disease advances and as it responds to treatment. Though her focus was on what was occurring inside the cell, her ultimate interest was on how these changes reverberated outside it, specifically on the immune system.
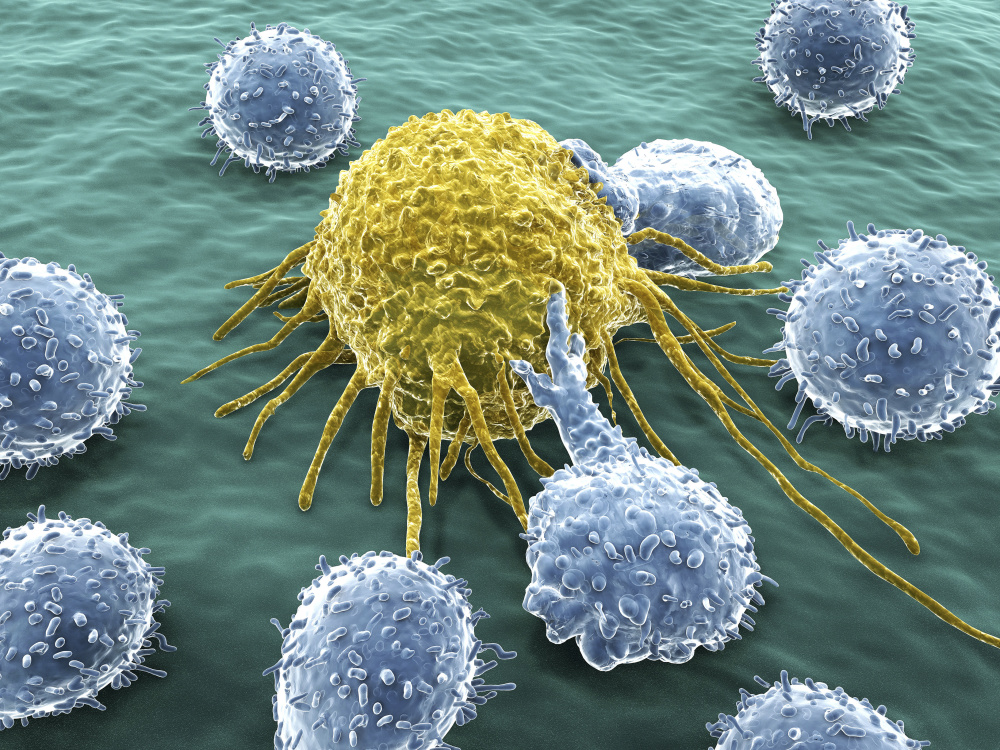
The human immune system exercises constant surveillance of the body’s tissues and cells. Like a conductor passing through a train car, immune system T cells inspect the ‘tickets’ of every cell they encounter to determine whether it is authorized to be there. Cells not so authorized — because they are foreign to the body or are infected or cancerous — are summarily killed. The ‘tickets’ take the form of antigens, bits of proteins arranged in strands that dangle from the cell surface and inform T cells whether the cell is normal and healthy — and therefore to be preserved — or diseased and defective, and to be destroyed.
While tumor cells share many of the same antigens as normal cells, they often betray their malignant nature by also displaying antigens unique to cancer cells. These so-called neoantigens are the stimulus that T cells need to launch an attack on any cell carrying them.
“Scientists are, as a rule, creatures of passion.”
Catherine Wu, MD
Neoantigens are the result of mutations to specific genes within cancer cells. The mutation causes the cell to make abnormal peptides, the pieces of proteins that form antigens. The exotic, eccentric, jerry-built neoantigen is then hoisted to the surface of the cell where it catches the immune system’s attention.
Wu and her lab team tracked the mutations that gave rise to specific neoantigens on the CLL cell surface.
“Through the early part of my career, we developed expertise in using antibodies and T cell-based techniques to try to discover what those antigens were,” Wu comments.
They were making steady progress when, in the early 2000s, a series of technological and computational advances emerged that accelerated their work and made it feasible to contemplate an entirely new form of immunotherapy for cancer.
First, next-generation sequencing provided researchers with a running tally of every letter of the DNA code within the nucleus of CLL cells. By focusing just on the sections of DNA that hold the blueprints for cell proteins, Wu and her colleagues could locate every mutation responsible for the peptides that comprise neoantigens.
The next step was to determine which neoantigens present the strongest lure to the immune system. To be “seen” by T cells, neoantigens must be framed within structures called HLA complexes, which serve as display cases for proteins from the cell surface. Advances in computer algorithms enabled researchers to determine, for each patient, which neoantigens were most likely to be presented on HLA complexes.
The elements were now in place for a personalized cancer vaccine, which Wu and her colleagues at Dana-Farber, the Broad Institute of MIT and Harvard introduced in 2016. Known as NeoVax, it is created by taking a sample of a patient’s tumor tissue and blood and sequencing their exomes — the sections of DNA that prescribe the proteins to be made by the cell.
By comparing the exomes of the tumor tissue and the patient’s normal tissue, scientists can identify the mutations that generate neoantigens. Then, using computer algorithms such as NetMHC, they predict which neoantigens are most likely to bind to MHC complexes.
They could then partner with companies to synthesize the peptides that form these especially presentable antigens, combine them with a stimulant, and inject them into the patient. The neoantigens spur the patient’s T cells to target the neoantigens on tumor cells.
(Ideally, Wu says, a vaccine would contain all the neoantigens found by sequencing the tumor DNA, but for practical reasons NeoVax uses a smaller number, usually between 15 and 20.)
In 2017, Wu and colleagues at Dana-Farber reported the results of the first clinical trial of NeoVax, in which the drug was tested in six patients with melanoma whose tumors had been surgically removed and who were considered at high risk for recurrence. At a median period of two years after vaccination, four of the patients had no evidence of cancer recurrence. In the two other patients, whose cancer had spread to their lungs by the time they were vaccinated, the disease recurred. After treatment with a drug known as an immune checkpoint inhibitor, the two patients had a complete remission of their tumors, demonstrating the potent synergy between the vaccine and immune checkpoint inhibitors.
Although it involved a small number of patients, the trial offered a “proof of principle” that neoantigen vaccines can stimulate a safe, potent, and highly targeted anti-tumor response in patients with melanoma, Wu remarks.
The melanoma trial was followed by a Dana-Farber-led trial involving eight patients with glioblastoma, an aggressive form of brain cancer. Although all the participants eventually succumbed to their disease, the cancer grew more slowly, and the patients lived longer, than has historically been the case for glioblastoma.
The trial contained other positives as well. One was that it demonstrated that NeoVax could successfully be made for cancers, like glioblastoma, that have relatively small numbers of mutations. (Tumors with few mutations tend to generate fewer of the neoantigens on which NeoVax is based.) Another was that researchers found evidence that T cells exposed to NeoVax zeroed in on tumor cells that carried the targeted neoantigens.
“It told us that even though we’re vaccinating peripherally [outside the brain], T cells with the right specificity are getting into the tumor,” Wu comments.
Encouraged by these results, Dana-Farber investigators have opened or are planning an array of follow-up trials. Toni Choueiri, MD, is leading a trial of NeoVax for patients with renal cell cancer. Patrick Ott, MD, PhD, who led the initial trial in melanoma, will start another trial of NeoVax in patients with metastatic melanoma later this year; and David Reardon, MD, who led the initial glioblastoma trial, will launch a trial that combines NeoVax with an immune checkpoint inhibitor in patients with glioblastoma.
The impulse to take the lessons of the initial trials back to the laboratory and then into a further round of trials is central to Wu’s approach to science.
“We’re always thinking about what we can learn from our patients. In the lab, we try to dissect the trial results — to identify general principles that we can then apply to the next generation of trials,” she relates. “It’s really a very tight cycle between clinical trials, laboratory investigation, and next-phase clinical trials.”

Forging a team
Wu’s acumen as a scientist is matched by her ability to lead a large and complex research undertaking, colleagues say. The number of researchers and the range of disciplines involved in translating basic research discoveries — about the intricacies of the immune response in stem cell transplants — into a personalized cancer vaccine is daunting under any circumstances. Wu conveys a belief not only in the promise of the research but also of the power of collaboration.
“Cathy is very skilled at assembling a multidisciplinary group of investigators who can approach a problem from different angles and work together toward a solution,” says Dana-Farber’s Margaret Shipp, MD, who has worked with Wu on a variety of research projects in lymphoid malignancies. “She doesn’t give up; she continues to work in an area until there’s clear success.”
She’s also impressive as a mentor to young investigators, Shipp continues. “She’s very committed to the junior members of the team — concerned about helping them succeed and mature in their careers.”
Wu excels in both the organizational and motivational aspects of leadership, colleagues say.
“She doesn’t just direct people, she gets people excited. She knows how to reach people when needed, and people respond to her,” comments Dana-Farber’s Patrick Ott, MD, PhD, who worked with her on the melanoma trial of NeoVax. “She’s an incredibly hard worker. She makes people want to work with her because they know she’ll ensure successful completion and a high impact of the collaborative project.”
It was in recognition of these skills that Wu was recently named chief of the new Stem Cell Transplant and Cellular Therapies Division at Dana-Farber.
“The creation of the division reflects the sea change that has occurred in our field in the past few years,” Wu remarks. “We’re envisioning it as a hub for immunotherapy across all different types of cancer. The foundation that stem cell transplants provided for this work has been critical. It’s a source from which we can continue to extract those critical ingredients that point the way to new therapies.”

I have small cell cancer, which is a death sentence. I was recently told at MA General Hospital, that immunotherapy may extend my life to the possibility of 12 to 18ths. I was not going to try chemo alone, I initially was choosing to stop all help and just succumbe to this disease. I was given hope with the immunotherapy possibilities. I know it’s not like 3 to 5 years, but I would like to be included in any trials once the immunotherapy runs out of helping me/ So far I have only had 1 cycle of immunotherapy and chemotherapy. I have no unreasonable side effects to date. I am currently being treated at the So Shore Dana Farber Cancer Institute. However, the Dr. I see there doesn’t see much hope, he said maybe extend my life 2 months. I was pretty discouraged. Is there anything else I can do or anyone else I can see that is really impressed with immunotherapy and sees hope ahead for this evolving new drug.
Thank you for reading. Due to the high volume of comments we receive, we are usually unable to respond to each person individually. If you have a specific medical question related to this blog post, we would recommend talking to your doctor or other care provider. We’ve also gathered answers to some of the more frequent questions below:
-For more information on a particular type of cancer or the latest updates we have available, please visit our website (https://www.dana-farber.org/) or search our blog by clicking the magnifying glass at the top of our homepage: https://blog.dana-farber.org/insight/
-For information on whether you would be eligible for a certain treatment, please visit our website for more information on how to make an appointment or get an online second opinion: https://www.dana-farber.org/appointments-and-second-opinions/
-For information on clinical trials available at Dana-Farber and elsewhere, please visit the Dana-Farber database: https://www.dana-farber.org/research/clinical-trials/find-a-clinical-trial/. For clinical trials outside of Dana-Farber: http://www.clinicaltrials.gov.
Wishing you all the best,
Dana-Farber Insight team
What markers exist on Sarcomas that a vaccine can help the immune system recognize the cancer? What kinds of genomic testing should one ask their doctor(s) to perform on a removed sarcoma tumor to see if there is a anti-cancer vaccine that might help in the treatment?
Great article!!! Keep up the good work.
I have a Stage 4 sarcoma, Giant Cell Sarcoma undifferentiated Soft parts.