For many patients with lymphoma or multiple myeloma, a stem cell transplant with their own stem cells (an autologous transplant) or CAR T-cell therapy can extend life significantly or even cure the disease. A variety of factors influence which of these two treatments is recommended, including:
- whether a patient is responding to chemotherapy
- whether the patient is likely to relapse after chemotherapy
- whether the patient has health conditions that make them ineligible for a transplant
What is the difference between stem cell transplant and CAR T-cell therapy?
An autologous stem cell transplant involves treatment with high doses of chemotherapy followed by an infusion of the patient’s own hematopoietic stem cells to rebuild their blood supply. It is generally effective only for patients who have responded to cancer therapies like chemotherapy and are in a near or complete remission.
CAR T-cell therapy, by contrast, involves collecting a different set of cells — the patient’s immune system T cells — and genetically modifying them to better recognize cancer cells. The modified cells are then injected back into the patient, where they latch onto and kill tumor cells.
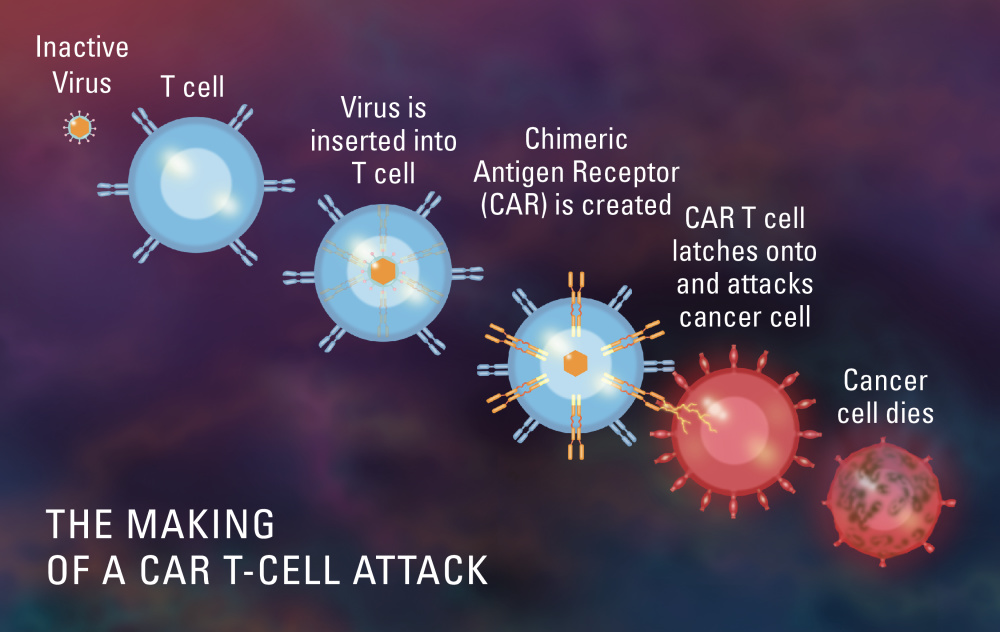
Before the CAR T cells are reinfused, patients may receive chemotherapy for their cancer, which helps create space in their immune system for the cells to expand and proliferate. In contrast to autologous stem cell transplantation, CAR T-cell therapy has been tested only in patients whose cancer has not entered remission from prior therapies.
The decision of which approach to take rests on several considerations:
- Patients whose lymphoma or multiple myeloma is resistant to their most recent chemotherapy regimen are not candidates for an autologous stem cell transplant. This is because an autologous transplant is essentially treatment with high-dose chemotherapy, given in hopes of overcoming the resistance of cancer cells not killed by standard-dose chemotherapy, and thus preventing a relapse. The transplanted stem cells are principally meant to “rescue” patients from the toxic side effects of the high doses of chemotherapy used to kill cancer cells and are not part of the anti-cancer therapy itself.
- Based on the results of several clinical trials, patients with lymphoma that is resistant to chemotherapy are eligible for a CAR T-cell therapy. CAR T-cell therapy was initially approved for patients whose disease had relapsed multiple times, but its success in those patients suggested it could be more effective than some of the standard therapies used in earlier lines of treatment. Two recent trials involved patients with B-cell lymphoma that didn’t respond to first-line treatment or relapsed within 12 months of that treatment. The trials, which compared CAR T-cell therapy to standard second-line therapy followed by an autologous stem cell transplant in responding patients, found CAR T-cell therapy to be more effective for these patients and is now available to them as a second-line therapy.
- For patients with multiple myeloma, CAR T-cell therapy is approved as a second-line treatment. That is, it’s offered as standard therapy only if patients haven’t benefited, or no longer benefit, from one previous course of treatment.
- Patients who are above age 75 or have heart disease, kidney disease, lung disease, or other health conditions not compatible with high-dose chemotherapy generally are not candidates for an autologous transplant. Many of these patients, however, are likely to be eligible for CAR T-cell therapy, as the toxicities and side effects of, and ability to tolerate, each treatment differs.
Patients with questions about stem cell transplant or CAR T-cell therapy should speak with their care team. Learn more about CAR T-cell therapy and stem cell transplants from Dana-Farber.
About the Medical Reviewer
 Caron A. Jacobson, MD, MMSc, is the Medical Director of the Immune Effector Cell Therapy Program at Dana-Farber Cancer Institute, which houses the CAR T-cell program at Dana-Farber/Brigham and Women’s Hospital, among other cellular therapies. Her area of clinical practice is in lymphoma, and her research interests and experience are in immunotherapies for the treatment of B-cell non-Hodgkin lymphoma.
Caron A. Jacobson, MD, MMSc, is the Medical Director of the Immune Effector Cell Therapy Program at Dana-Farber Cancer Institute, which houses the CAR T-cell program at Dana-Farber/Brigham and Women’s Hospital, among other cellular therapies. Her area of clinical practice is in lymphoma, and her research interests and experience are in immunotherapies for the treatment of B-cell non-Hodgkin lymphoma.
I had a CAR T cell therapy procedure in 2021 for Multiple Myeloma in a clinical trial. Prior to that I had a failed Stem Cell transplant in 2019. To date I am still in remission.
Enjoyed your well written article and forwarded it to my family for more clarity in their knowledge of this form of treatment.
Six months out from my CAR-T for multiple myeloma and I haven’t felt this good in seven years. Nothing else was working and I wasn’t tolerating chemo well any longer. The CAR-T was life changing. Not hard to recover from. Completely different recovery from my stem cell transplant. Much much easier. I’m obviously nervous about when it will stop working but in the meantime I am enjoying life off of any other meds except twice daily acyclovir. Joint inflammation is a side effect (especially my hands and knees) but I’ll take it!
Thank you for this article helping me understand newer available Lymphoma treatments and there differences.
My mother and sister had Lymphoma. (age 90 yrs and 68 yrs)
I try to stay aware of new research about this challenging immune disease and read all Farber articles sent via email.
Thanks. You offer caring and hope to all.
Blessings in all you do.
I had CAR-T cell therapy in 2021 and I can testify this is the best thing that could have happened to me I feel like a new person the procedure was so productive for me that I recommend anyone that qualifies for it to do so it wa a life changing treatment for me now I am able to live normal again I just want to thank my doctor Eric Jacobsen for all what he done for me and Dana Farber cancers team